Glucose management
Care300® is a patented organic standardised mango fruit powder (Mangifera indica spp Totapuri). Care300® activates the key metabolic enzymes SIRT1 and AMPK, improving metabolism and glucose tolerance. Supported with 1 published clinical trial. 300 mg/day.
Care300® - Key features
-
Patented ingredient
-
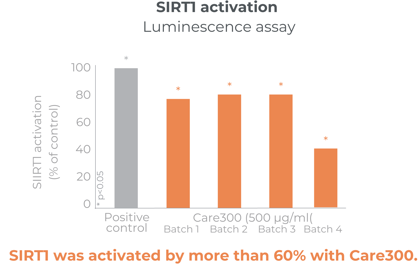
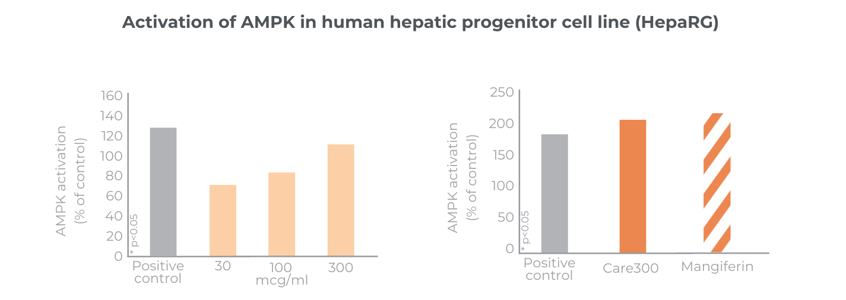
Plays a role in the activation of SIRT1 and AMPK for glucose uptake
-
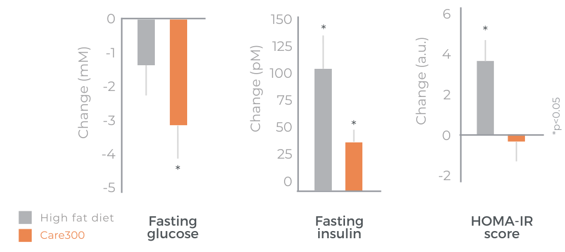
Efficient on insulin sensitivity
-
Protects metabolism homeostasis (SOD, inflammatory markers)
-
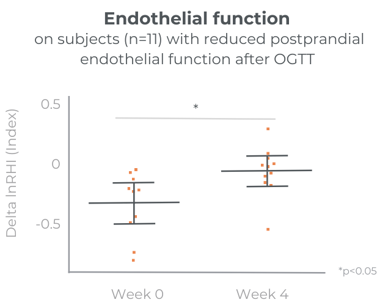
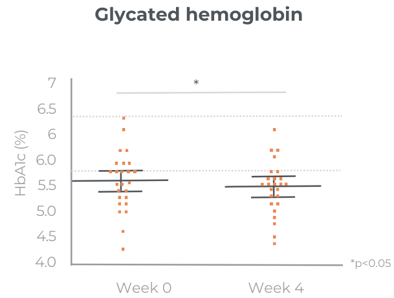
Significant improvement in glucose levels after 1 month
Science supporting Care300®
Care300® improves glucose management. Its mechanism of action in SIRT1 and AMPK modulation has been analysed in in-vitro studies. Its benefits have been confirmed in a chronic gold-standard clinical study.
Regulatory & Certifications
Organic certified
Non-GMO, Halal, Kosher, gluten-free, suitable for vegetarians
References
MECHANISTIC STUDIES
Röchter S. et al. Agro Food Industry Hi-Tech. 2015; Vol. 26(3)
Gerstgrasser et al., Planta Med, 2015; 82(4), 298-304
Sabater G. A. et al. Cell Physiol Biochem. 2017; 42:564-578
CLINICAL TRIALS
Buchwald-Werner S. et al. Planta Med. 2017; 83(10): 824-829
This website is intended to provide information about Fytexia’s ingredients, used in various food/dietary supplement products around the world. It is only intended for business to business and to provide information to food/dietary supplement professionals and is not designed for the general public. Statements used on this website have not been evaluated by the Food and Drug Administration or any other competent authority. Products are not intended to diagnose, treat, cure or prevent any disease.